Irregular pattern of fibrosis
Case #1:
Clinical History and Presentation
This is a 63-year-old patient with a significant past medical history of right ventricular failure, atrioventricular block status post dual-chamber pacemaker placement, patent foramen ovale (PFO) closure, atrial myxoma status post surgical resection in 2012, and suspected cirrhosis.
The patient presented to the liver clinic with marked fluid overload and was directly admitted for further evaluation. Prior liver function tests (01/13/2026) were within normal limits. Hemodynamic assessment revealed an elevated wedge pressure of 32 mmHg and a right atrial pressure of 29 mmHg. Given these findings and the concern for underlying liver disease, the patient underwent transjugular liver biopsies from both the right and left hepatic lobes for further diagnostic evaluation.
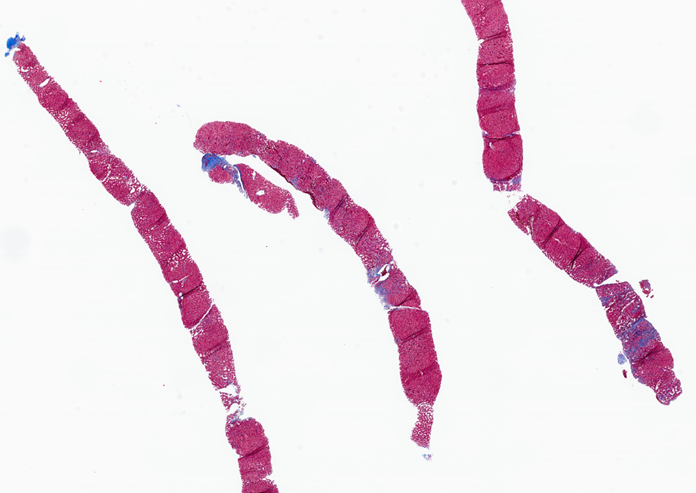
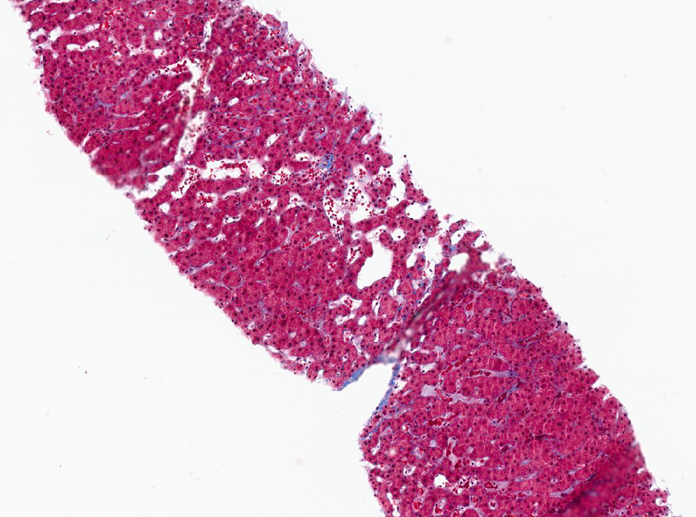
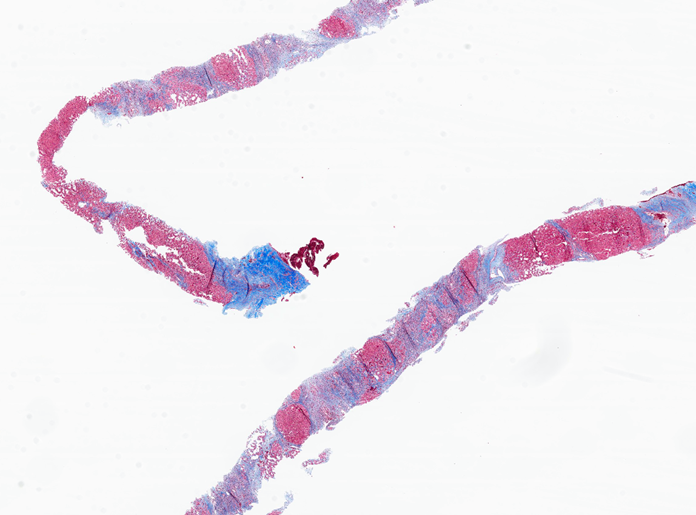
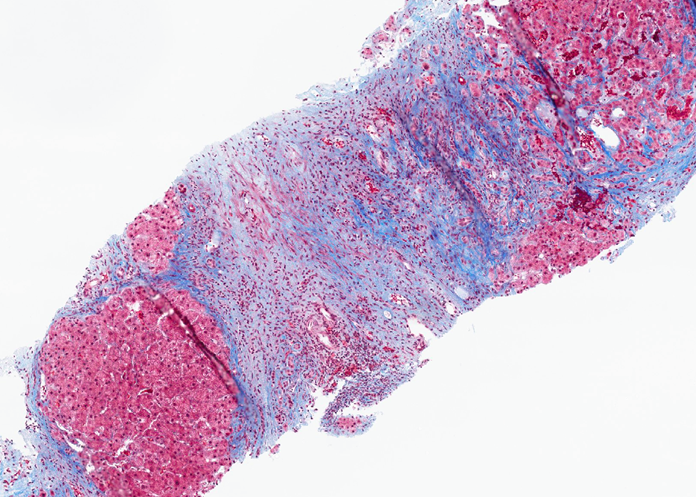
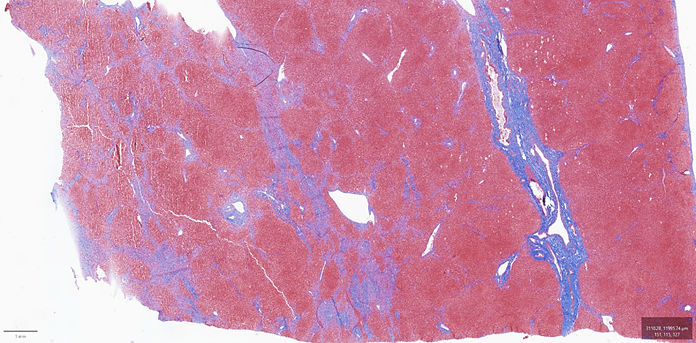
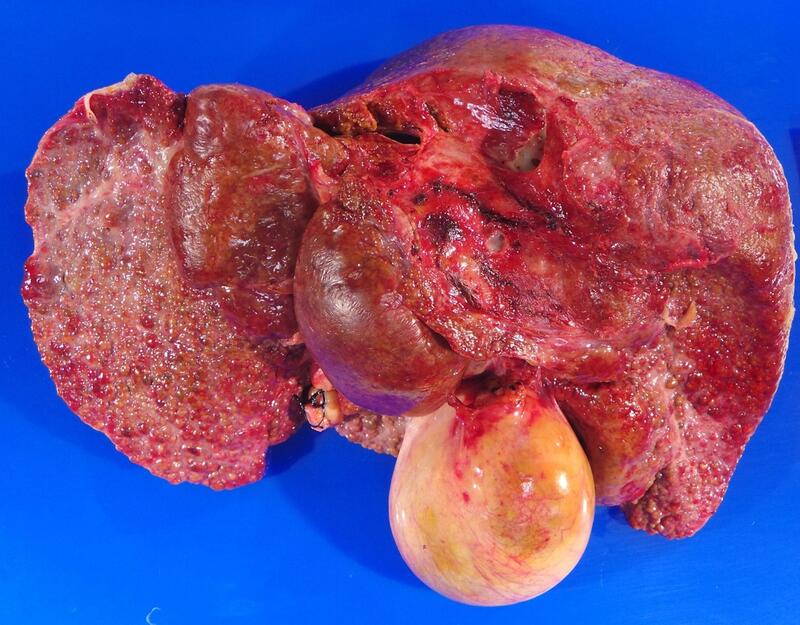
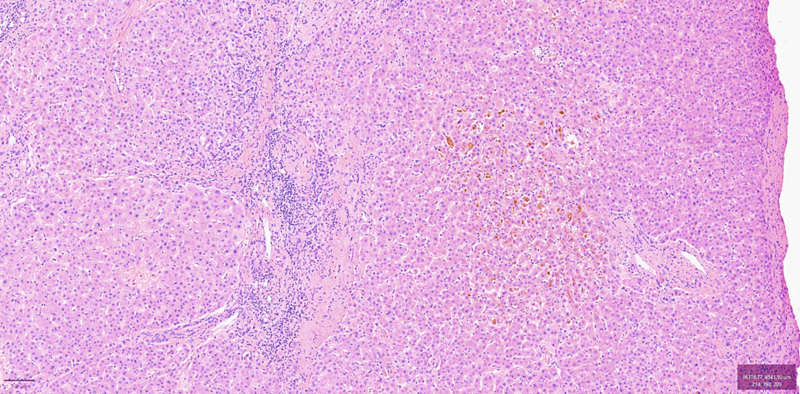
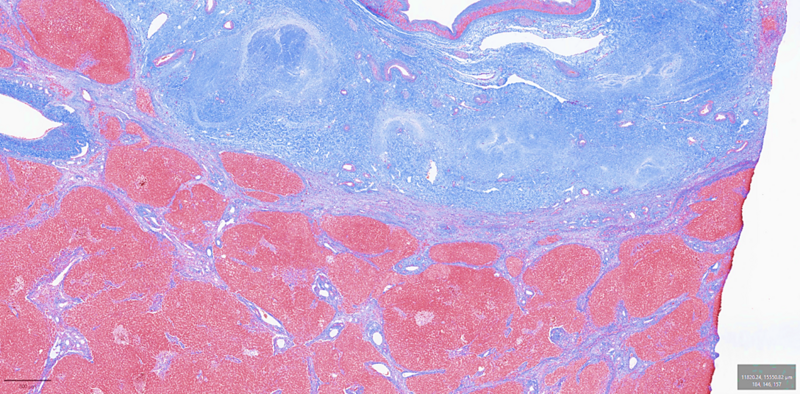
The right lobe liver biopsy on trichrome stain reveals patchy sinusoidal fibrosis with an overall lack of fibrosis in the majority of the cores (a). A higher-power view of one of the right lobe cores reveals only limited sinusoidal fibrosis (b). In contrast, the left lobe liver biopsy shows extensive fibrosis highlighted by the trichrome stain, with nodular formations characteristic of cirrhosis (c). The higher-power view of the left lobe cores highlights regenerative nodule formation (d). A representative section of the explant reveals prominent irregular fibrosis that is more pronounced on the left side compared to the right side of figure (e).
Discussion:
Cirrhosis can arise from chronic obstruction of hepatic venous outflow, as occurs in patients with long-standing congenital heart disease or other causes of chronic right-sided heart failure that lead to persistent venous backflow and sustained hepatic congestion. In this context, the liver injury is commonly referred to as congestive hepatopathy with advanced fibrosis, or cardiac cirrhosis.
Elevated central venous pressure and/or reduced cardiac output cause sinusoidal dilatation and congestion, centrilobular hypoxia, and hepatocyte damage. Over time, these changes can progress from centrilobular sinusoidal fibrosis to an irregular, patchy pattern of fibrosis that ultimately results in cirrhosis. Histologically, early findings include sinusoidal dilation and congestion, hepatocyte atrophy, variable hemorrhage predominantly in zone 3, and focal centrilobular necrosis. These are followed by collagen deposition in the centrilobular areas, which then extends outward to form bridging fibrosis between central veins and portal tracts.
Even in advanced stages, the fibrosis often remains highly heterogeneous: some regions of the liver show fully developed cirrhosis, while others display only mild or limited fibrosis. As a result, liver-related symptoms may be subtle, and the overall clinical picture is usually dominated by the underlying cardiac condition, such as valvular disease, severe pulmonary hypertension, cor pulmonale, biventricular heart failure, pericardial disorders, cardiac tamponade, or constrictive pericarditis.
Macroscopically, the liver is typically markedly enlarged and congested. Because of the heterogeneous nature of fibrosis, a single biopsy sample may underestimate the true stage of disease. Therefore, obtaining tissue from both the right and left hepatic lobes is recommended to enhance diagnostic accuracy and better capture the overall extent of fibrosis.
Management is directed primarily at optimizing cardiac function, as improvement in hemodynamics can lead to stabilization or partial regression of hepatic injury. In patients with advanced hepatic fibrosis or established cardiac cirrhosis, combined heart–liver transplantation may be considered.
Case #2:
Clinical History and Presentation
This is a 41-year-old patient with a known diagnosis of primary sclerosing cholangitis (PSC), scheduled for orthotopic liver transplantation on 10/21/2025. Recent liver function tests (10/09/2025) show a cholestatic pattern of liver injury with elevated total bilirubin (2.4 mg/dL), alkaline phosphatase (271 U/L), AST (78 U/L), and ALT (76 U/L). The patient is seronegative for viral hepatitis A, B, and C. ERCP reveals multifocal stricturing and dilatation of the intrahepatic and extrahepatic bile ducts, including a 3.6 cm segment of narrowing in the mid to distal common bile duct, in keeping with the classic “beading” appearance of PSC and advanced biliary stricturing necessitating transplantation.
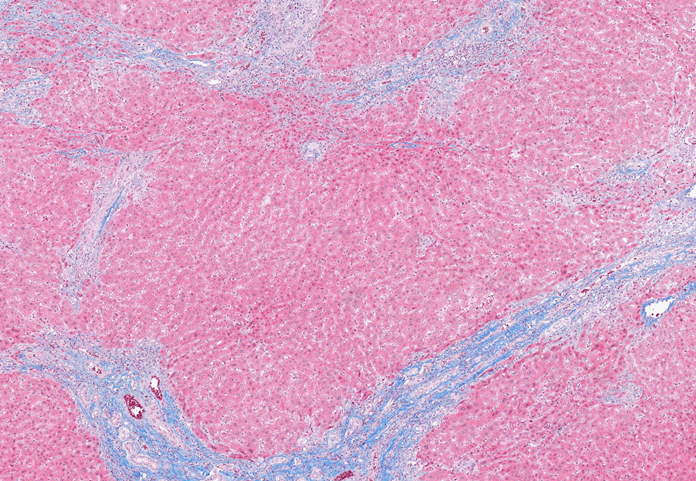
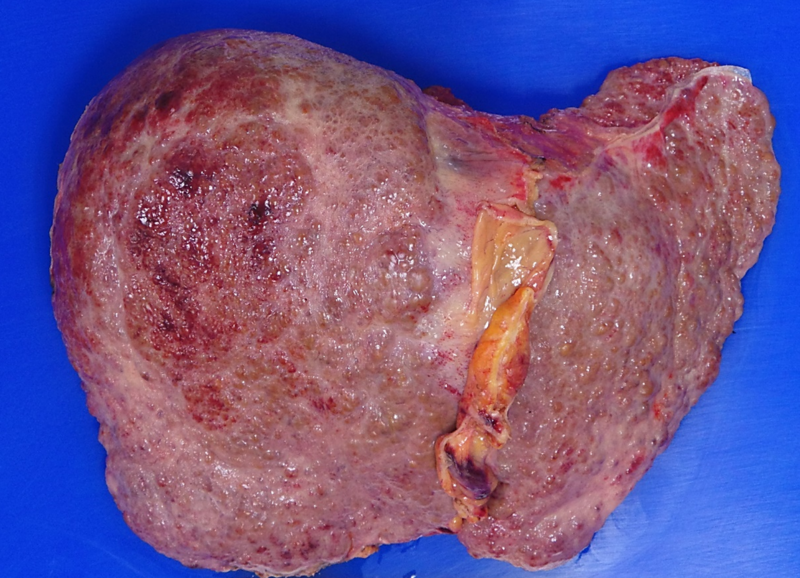
A macroscopic picture of the explanted liver from a patient with PSC reveals a nodular architecture (a). A representative section of this explant demonstrates the lobular parenchyma with notable moderate hepatocellular and canalicular cholestasis (b). The trichrome stain illustrates several native bile ducts completely replaced by fibrous scars (fibro-obliterative lesions). Additionally, it reveals an irregular pattern of fibrosis with areas showing limited to no fibrosis (c). A higher-power view shows an area with bridging fibrosis and early features of nodular formation (d).
Discussion:
Biliary cirrhosis represents the end result of prolonged cholestatic injury to the liver, whether from primary or secondary biliary disease. In clinical practice, it is most often encountered as a sequela of chronic obstructive cholangiopathies, particularly primary sclerosing cholangitis (PSC). The common pathogenic thread in these conditions is persistent impairment of bile flow, which leads to progressive portal-based fibrosis, distortion of the normal lobular architecture, and ultimately cirrhosis.
The progression to cirrhosis in biliary disease is highly variable and reflects several factors, including the etiology and level of obstruction and the presence of superimposed infection. A striking contrast is seen between infants and adults. In infants with unrelieved extrahepatic obstruction, such as biliary atresia, the liver progresses rapidly to cirrhosis, often within 5–6 months. By contrast, in adults, biliary cirrhosis usually develops only after many years of ongoing biliary disease, consistent with a more insidious, chronic course of injury.
Macroscopic changes in the liver mirror this evolution. In earlier stages, the liver tends to be enlarged as a result of inflammation, cholestasis, and early fibrotic change. With time, as established cirrhosis supervenes, the organ becomes shrunken to subnormal size. The external surface may appear smooth, finely granular, or frankly nodular, reflecting variable degrees of regenerative nodule formation and fibrosis. On cut section, the parenchyma typically shows a yellow to greenish-grey or deep green discoloration due to retained bile. Enlarged, fibrotic portal tracts stand out as small grey or white foci, and the intrahepatic bile ducts may be markedly dilated, containing light or dark bile that is sometimes inspissated.
Histologically, the appearance of biliary cirrhosis depends on the stage of disease and is often heterogeneous within the same liver. A key feature is cholestasis, which can be diffuse or predominantly periseptal, outlining the periphery of irregularly shaped regenerative nodules. This distribution underscores the portal-based nature of the injury. There is also considerable variation in the size and number of cirrhotic nodules in different regions, a point of practical importance because a small biopsy may sample only a limited and unrepresentative area, making assessment of the full extent of cirrhosis challenging.
Several characteristic microscopic lesions reflect the toxic and mechanical effects of retained bile. Feathery degeneration of hepatocytes results from intracellular accumulation of bile salts, imparting a swollen, finely vacuolated appearance to the cytoplasm. Focal bile infarcts represent areas of hepatocellular necrosis induced by concentrated bile, while bile lakes arise when bile escapes from damaged canaliculi or ducts into the surrounding parenchyma, forming pools of bile often accompanied by an inflammatory and fibrotic reaction. Together, these gross and microscopic changes illustrate how chronic biliary obstruction culminates in a distinctive form of cirrhosis, in which classical features of end-stage liver disease coexist with prominent cholestatic pathology.
Case #3:
Clinical History and Presentation
A 62-year-old male with a known history of metabolic dysfunction–associated alcohol-related liver disease (MetALD) cirrhosis had maintained complete abstinence from alcohol for the past five years. Liver function tests showed: ALP 165 U/L, AST 34 U/L, and ALT 28 U/L. The patient was scheduled for, and subsequently underwent, an orthotopic liver transplantation without any reported intraoperative complications.
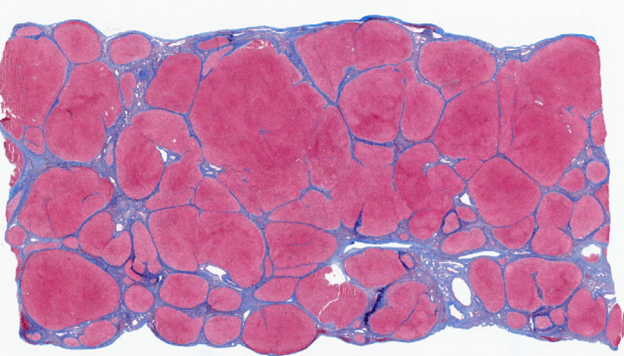
A macroscopic picture of the explanted liver from a patient with MetALD reveals a nodular architecture (a). A trichrome-stained section of the explant shows predominantly large macronodules separated by incomplete fibrous septa (b). In several areas, the fibrous bands appear thinned and display a curved, remodeled architecture, features consistent with regression and partial resolution of hepatic fibrosis. Overall, the architectural pattern is irregular and differs from the more uniform appearance typically seen in well-established cirrhosis due to MASH or alcohol-related liver disease (ALD).
Discussion:
For many years, cirrhosis was considered the irreversible end stage of chronic liver disease. It was once taught that once cirrhosis develops, the process is essentially one-way, with a focus on complications and transplantation rather than regression. However, current evidence has shifted this view. We now understand that advanced fibrosis, and even established cirrhosis in some patients, can regress when the underlying injurious process is effectively treated or removed.
Historically, this concept emerged from case reports and small series in which patients with well-documented cirrhosis showed improvement after successful therapy. In some of these patients, “full-blown” cirrhosis evolved into incomplete septal cirrhosis, while in others there was an apparent disappearance of fibrosis on follow-up biopsy. These early observations were reported in conditions such as hereditary haemochromatosis treated with iron removal, autoimmune hepatitis controlled with immunosuppression, Wilson disease managed with copper chelation or zinc therapy, and extrahepatic biliary obstruction relieved by restoring bile flow. Although initially regarded as rare exceptions, these cases challenged the dogma that cirrhosis is always irreversible.
More recently, evidence from larger cohorts has confirmed that fibrosis and even cirrhosis can regress in a substantial proportion of patients once the driver of liver injury is controlled. In chronic hepatitis B and C, effective antiviral therapy has been associated with clear improvements in fibrosis stage and documented regression of cirrhosis on serial biopsies or non-invasive assessment. Similarly, in steatohepatitis, addressing the causative factors through healthier diets, weight loss, metabolic control, and cessation of alcohol can lead to reduced inflammation and gradual remodelling of fibrotic tissue.
It is important to recognize that regression of fibrosis is typically a slow process, often requiring years of sustained control of the underlying disease. The time course and extent of regression depend on several factors, including the etiology of liver disease, the severity and duration of injury at the time of intervention, and how completely the injurious stimulus is removed. Even when there is histologic regression of cirrhosis, the liver may not fully return to normal; architectural and vascular changes can persist, and the risk of hepatocellular carcinoma or portal hypertension may remain elevated.
References
- Burt, MacSween's Pathology of the Liver: Eighth Edition. 2022.
- Wells ML, Fenstad ER, Poterucha JT, Hough DM, Young PM, Araoz PA, Ehman RL, Venkatesh SK. Imaging Findings of Congestive Hepatopathy. Radiographics. 2016 Jul-Aug;36(4):1024-37. doi: 10.1148/rg.2016150207. Epub 2016 Jun 10. PMID: 27284758.
- Hollenberg SM, Waldman B. The Circulatory System in Liver Disease. Crit Care Clin. 2016 Jul;32(3):331-42.
- Koehne de Gonzalez AK, Lefkowitch JH. Heart Disease and the Liver: Pathologic Evaluation. Gastroenterol Clin North Am. 2017 Jun;46(2):421-435. doi: 10.1016/j.gtc.2017.01.012. PMID: 28506373.
- Wanless IR, Nakashima E, Sherman M. Regression of human cirrhosis: morphologic features and the genesis of incomplete septal cirrhosis. Arch Pathol Lab Med. 2000;124:1599–1607.
- Issa R, Zhou X, Constandinou CM, et al. Spontaneous recovery from micronodular cirrhosis: evidence for incomplete resolution associated with matrix cross-linking. Gastroenterology. 2004;126:1795–1808.
- Quaglia A, Alves V, Balabaud C, et al. Role of aetiology in the progression, regression, and parenchymal remodelling of liver disease: implications for liver biopsy interpretation. The International Liver Pathology Study Group (2016) Histopathology68, 953–967.
- Hytiroglou P, Theise ND. Regression of human cirrhosis: an update, 18 years after the pioneering article by Wanless et al. Virchows Arch. 2018;473:15–22.