Why do we transplant some types of cholangiocarcinoma and not others?
Cholangiocarcinoma: It’s not just one tumor
Cholangiocarcinoma (CCA) represents a heterogeneous group of malignancies arising from the biliary epithelium. It is divided into three subtypes depending on their anatomical site of origin: intrahepatic (iCCA), perihilar (pCCA) and distal (dCCA). iCCAs arise above the second-order bile ducts, while pCCA (also called a Klatskin tumor) arise above the cystic duct with dCCA coming from below the cystic duct. pCCA is the single largest group, accounting for approximately 50–60% of all CCAs, followed by dCCA (20–30%) and iCCA (10–20%).
In this post, we will primarily discuss pCCA and iCCA, as dCCA is treated surgically with pancreaticoduodenectomy (Whipple procedure) rather than liver transplantation.
CCA accounts for approximately 3% of all gastrointestinal cancers representing the second most common primary hepatic malignancy after hepatocellular carcinoma (HCC). Unlike HCC, the majority of CCA cases occur in the absence of an evident chronic liver disease or other risk factor making it more difficult to catch early. Primary risk factors are Primary Sclerosing Cholangitis (PSC), with a lifetime risk of 10%, rising to 30% in those with Inflammatory Bowel Disease (which is why we annually screen these patients with an MRI), biliary cystic disease, and less commonly hepatitis B. Outside of the United States, southeast Asian liver flukes remain a risk factor.
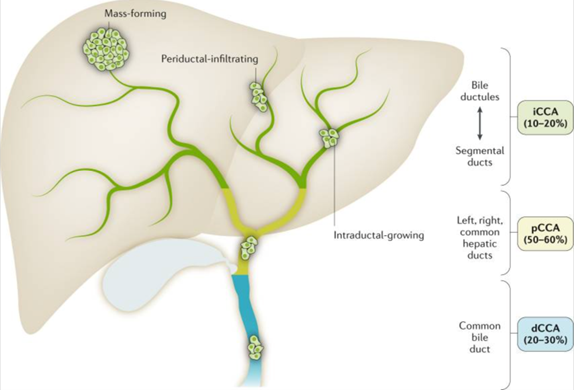
Figure 1: Types of cholangiocarcinoma with the incidence of intrahepatic cholangiocarcinoma 10-20% and the incidence of perihilar cholangiocarcinoma 20-30%
How do we diagnose CCA?
There are a few situations when to suspect CCA
- An asymptomatic rise in cholestatic liver enzymes or painless jaundice
- Symptoms of cholestasis or cholangitis with imaging findings concerning for malignancy
- A rise in liver enzymes (particularly Alkaline phosphatase and bilirubin) or CA19-9 in a patient with PSC
Unfortunately, unlike patients with dCCA and pCCA who present with biliary obstruction, those with iCCA may present with vague symptoms such as abdominal pain, weight loss and may not present until advanced stages.
An MRI of the Liver with Magnetic Resonance Cholangiopancreatography (MRCP) is the primary imaging modality, though iCCA can be difficult to differentiate from HCC. In fact, 0.7%–3.9% of liver explants from either resection or transplantation are ultimately found to represent iCCA or mixed tumors despite a preoperative diagnosis of HCC. When either a pCCA or an iCCA is suspected a CT chest should be done to rule out lung metastasis.
If this is a patient with PSC, patients should be screened for a dominant stricture on MRCP or elevated CA 19-9 (>20 U/ml or rising). A dominant stricture is defined by a diameter of ≤1.5 mm in the common bile duct or ≤1 mm in the hepatic duct. Although this does not always mean the patient has a malignancy, every dominant stricture must be investigated. The next step entails an ERCP with brushing, fluorescence in situ hybridization (FISH) and if possible directed biopsy with cholangioscopy.
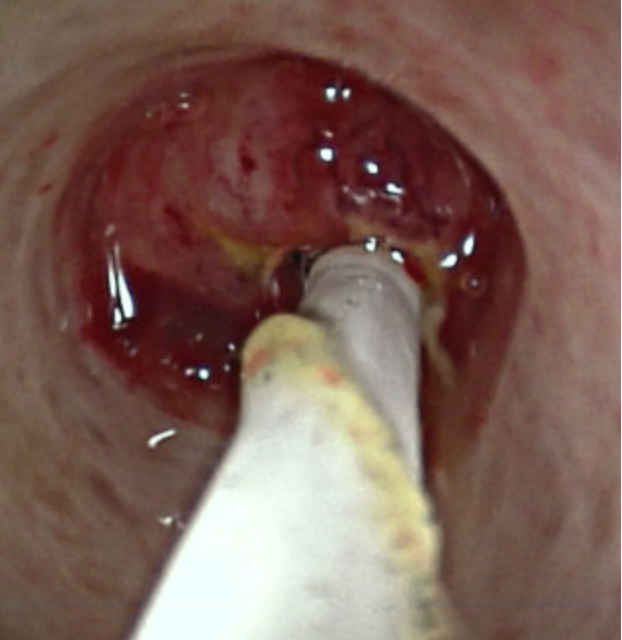
Figure 2: Cholangioscopy image of cholangiocarcinoma
In patients without PSC, the diagnosis can occur due to a lesion seen when screening a patient with cirrhosis, a more advanced lesion causing obstructive symptoms or less commonly incidentally found on imaging (Figure 3). In PSC, it can be found sooner as these patients are screened more frequently with an MRI (Figure 4).
Sometimes it is difficult to distinguish from an HCC when it is not clearly HCC by LI-RADS criteria. The preferred method of diagnosis is done via intraductal sampling, but this can sometimes be difficult if it is very peripheral necessitating a percutaneous biopsy. Additionally, if it is a hilar mass the guidelines suggest ERCP guided sampling with FISH or cholangioscopy if possible, but this doesn’t always yield a diagnosis. It is important that EUS-FNA/B or percutaneous biopsy is avoided especially in patient who may become transplant candidates as one study from the Mayo Clinic noted high rates of peritoneal metastasis (5 out of 6 patients). In fact, many transplant protocols consider prior transperitoneal sampling an absolute exclusion criterion. In these cases, EUS lymph node sampling and help make the diagnosis.
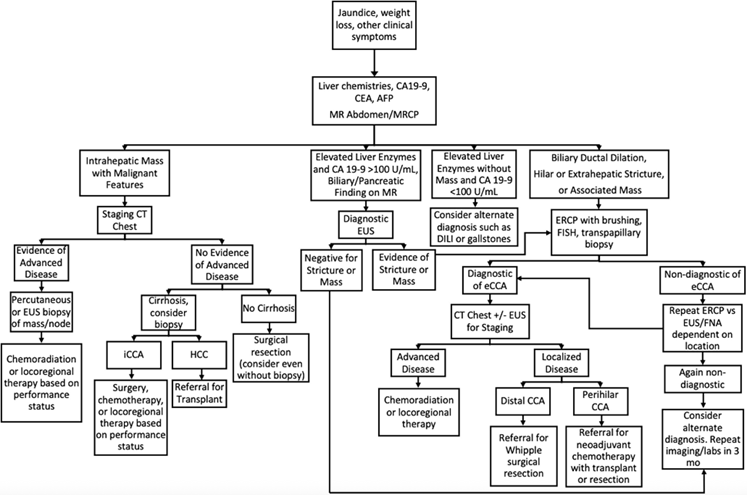
Figure 3: CCA diagnosis and management in a patient without PSC
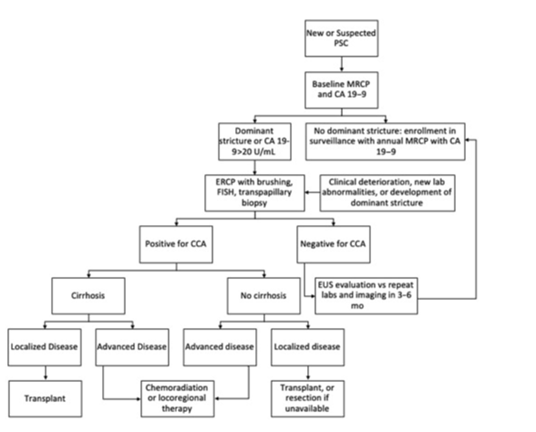
Figure 4: CCA diagnosis and management in a patient with PSC
What is the treatment paradigm for pCCA?
pCCA has a unique anatomic location at the hepatic hilum where the right and left hepatic ducts converge, near key vascular structures—the portal vein and hepatic artery. This anatomic complexity makes treatment selection challenging and necessitates multidisciplinary discussion given the significant ramifications of each approach.
Why surgical resection has limitations
For patients without underlying liver disease and with early-stage, resectable tumors, surgical resection may be attempted (criteria for which is outlined in Milan Consensus). However, outcomes are disappointing. A 2005 study of 53 patients who underwent curative-intent resection demonstrated median survival of only 40 months with 5-year survival of 35-45%, even when negative margins (R0 resection) were achieved.
The problem is two-fold: First, achieving negative margins requires extensive hepatectomy that many patients—particularly those with PSC and underlying cirrhosis—cannot tolerate due to insufficient future liver remnant. Second, even when technically successful, recurrence rates remain high because the entire biliary tree in PSC patients is diseased, making bile duct reconstruction problematic.
For unresectable disease, systemic therapy offers limited benefit. Gemcitabine/cisplatin plus durvalumab (based on TOPAZ-1 trial) yields median overall survival of only 15.1 months.
These poor outcomes with conventional approaches—limited resectability in PSC patients, high recurrence even after R0 resection, and dismal survival with systemic therapy—created the rationale for exploring liver transplantation as an alternative in carefully selected patients.
When do we transplant for pCCA and how did that come about?
The modern era of CCA transplantation came from the Mayo clinic where Dr. Gregory Gores and Dr. Charles Rosen developed a protocol for patients with unresectable pCCA.
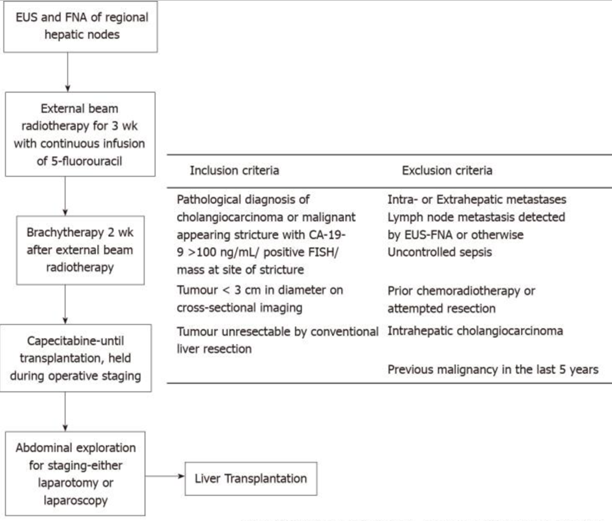
Figure 5: Mayo protocol for Liver transplantation
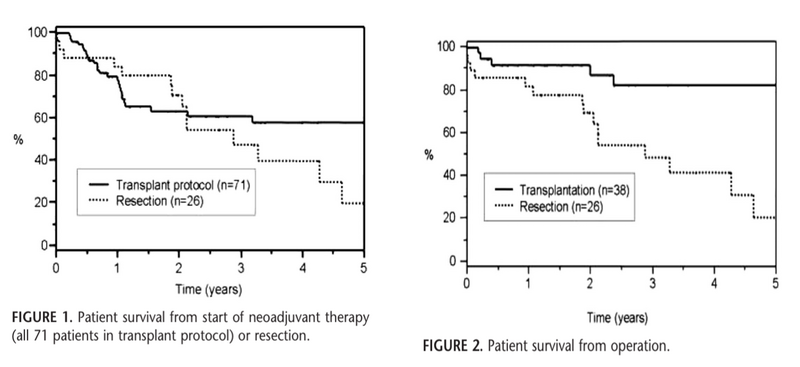
Their landmark 2005 publication reported outcomes from 1993-2004 in 71 patients with unresectable pCCA who entered the neoadjuvant protocol, with 38 ultimately proceeding to transplant. Compared to 26 patients who underwent surgical resection, transplant recipients demonstrated superior outcomes: 5-year survival of 82% versus 21% (p=0.022) and lower recurrence rates (13% vs. 27%). Importantly, outcomes were equivalent between PSC-associated and de novo pCCA, establishing that transplantation could benefit carefully selected patients regardless of underlying liver disease.
Figure 6: Survival of patients who underwent liver transplant for hilar cholangiocarcinoma
For the first time this reinforced that focusing on patients with favorable tumor biology and excluding those with progressive disease during neoadjuvant therapy can demonstrate excellent survival outcomes with liver transplant.
A 2012 update with multiple centers including 287 patients demonstrated 5-year intent-to-treat survival of 53% and 5-year post-transplant survival of 65%.
Changes in UNOS policy
The success of the Mayo protocol led to the approval by United Network for Organ Sharing (UNOS) in 2009 of a national pilot study allowing pCCA patients meeting specific criteria to receive MELD exception points, similar to HCC patients.
MELD Exception Points for pCCA
- Diagnosis of pCCA meeting one of the following diagnostic criteria
- ≤3 cm in radial diameter hilar mass
- Consistent biopsy or cytology results
- Aneuploidy via FISH
- CA 19-9 >100 without cholangitis.
- The mass must be unresectable because of technical considerations or due to underlying liver disease
- Completion of neoadjuvant therapy per an approved protocol
- Assessment of regional lymph nodes and peritoneal metastases by operative staging after completing neoadjuvant therapy. EUS can be performed based lymph node biopsy prior to neoadjuvant therapy. There can be no evidence of metastatic disease
- CA 19-9 ≤100 U/mL (or ≤1,000 U/mL with documented cholangitis)
- ≤3 cm in radial diameter hilar mass
Initially, the pilot study required underlying PSC as an eligibility criterion. This requirement reflected the observation that PSC-associated pCCA appeared to have more favorable tumor biology compared to de novo pCCA. However, subsequent data demonstrated that carefully selected patients with de novo pCCA could achieve similar outcomes.
Additionally, the staging laparoscopy has become an important part of this process, as some studies have demonstrated that 27% of patients with radiographically resectable pCC have occult peritoneal metastases or positive peritoneal cytology discovered at staging laparoscopy. The timing of staging laparoscopy is important. Some centers perform an initial staging laparoscopy before initiating neoadjuvant therapy to avoid exposing patients with metastatic disease to toxic treatment. OPTN policy is for patients to undergo staging laparoscopy prior to LT and completion of neoadjuvant therapy.
Early allocation policies assigned escalating MELD exception points (starting at 22 for pCCA, 28 for HCC), unintentionally prioritizing exception candidates over equally sick non-exception patients. The 2019 MMaT-3 policy (median MELD at transplant minus 3 after 6-month stability) corrected this inequity and dramatically improved pCCA outcomes: 1-year transplant probability increased from 56% to 74%, and 3-year waitlist mortality decreased from 27% to 18%.
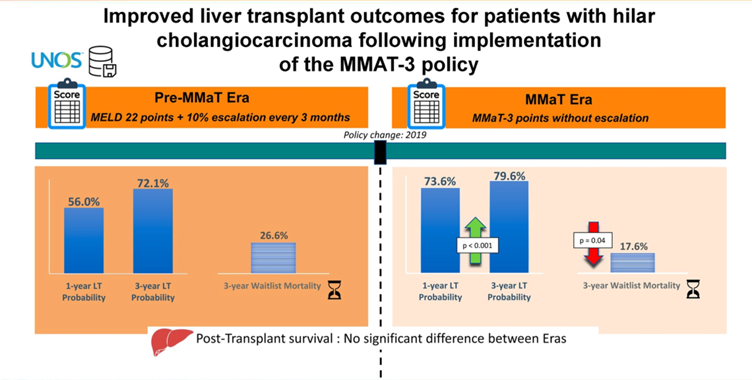
Figure 7: Changes to LT probability and waitlist mortality in the MMaT era
A recent meta-analysis demonstrated improved survival for pCCA, where 65% survived in those who completed neoadjuvant chemotherapy versus 31.6% who did not. This highlights the importance of adhering to the Mayo protocol.
What about iCCA and why does our transplantation approach differ?
As compared to pCCA, iCCA looked very different until recent years.
Liver resection is the treatment of choice for these tumors, although outcomes are poor with a median and 5-year OS of 28 months and 30%, respectively. Early attempts at LT for iCCA were uniformly disappointing. A 2000 multi-institutional study analyzed 207 patients who underwent LT for iCCA and pCCA, demonstrating median survival of only 23 months, and only 11 patients had a 5 year recurrence free survival. Tumor recurrence occurred in 51% of patients, with median time to recurrence of 8 months.
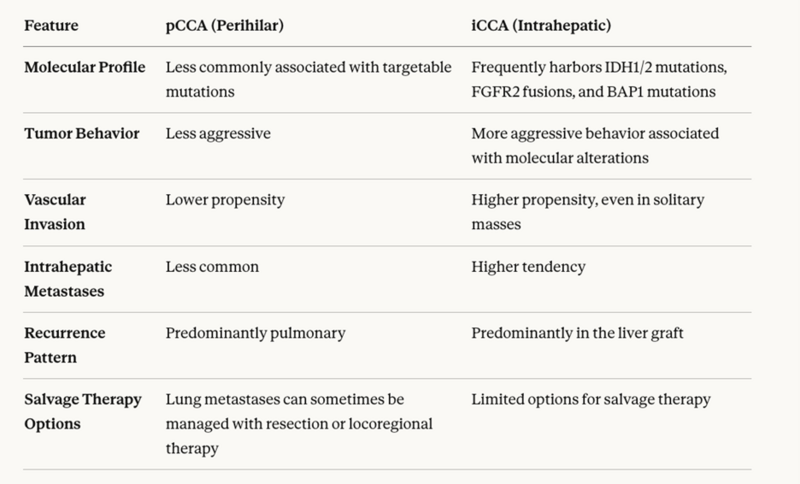
There are significant differences between pCCA and iCCA. iCCA demonstrates more aggressive biology (IDH1/2, FGFR2, BAP1 mutations) with higher rates of vascular invasion, intrahepatic metastases, and graft recurrence, limiting salvage therapy options.
Figure 8: Comparing pCCA and iCCA
Interestingly, iCCA can be misdiagnosed as HCC or the explant will show a mixed HCC/iCCA and after transplantation using the Milan Criteria. A 2014 study found that in patients with very early iCCA that were either found incidentally or were transplanted for HCC but found to have iCCA or a mixed tumor on explant of a size less than 2cm had a 5 year survival of 73%. This was expanded upon in a 2020 study that found that patients with cirrhosis with an iCCA between 2-5cm who had LT had a lower recurrence rate (21% vs. 48%) and a higher 5‐year RFS (74% vs. 40) compared to resection.
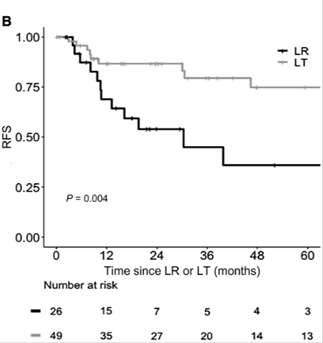
Figure 9: RFS for patients with iCCA comparing liver resection vs transplant
This ultimately led to a very recent change in 2025 that allows for exception points for iCCA if it meets the following criteria:
- Solitary tumor < 3 cm
- Unresectable and/or presence of cirrhosis
-
Stability of hepatic-only disease for ≥ 6 months
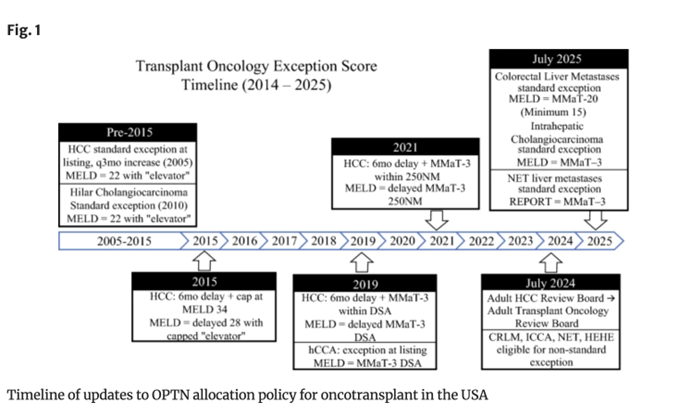
Figure 10: Timeline of Exception Point Policy changes
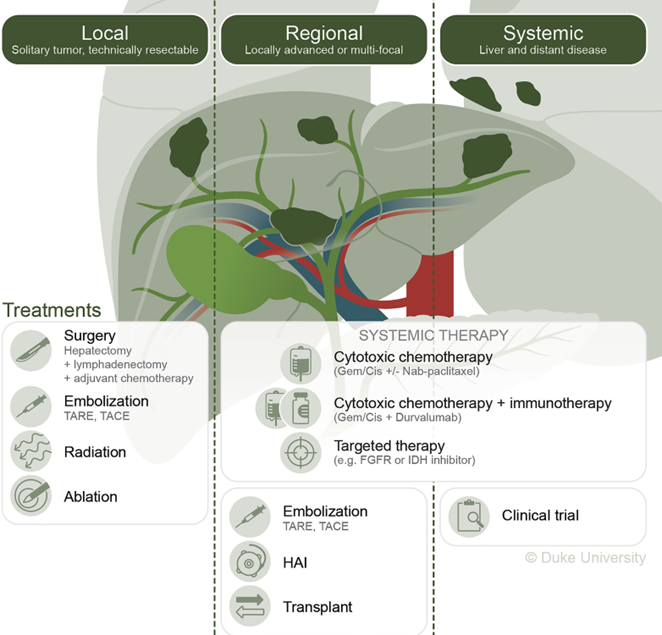
Figure 11: Treatment options for CCA
Take Home Points
- Intraductal sampling (ERCP brushings with FISH, cholangioscopy-directed biopsy) is preferred for diagnosis of pCCA in potential transplant candidates. EUS-FNA/B and percutaneous biopsies risk peritoneal seeding and are absolute contraindications to transplantation in most protocols.
- The Mayo Clinic protocol revolutionized pCCA treatment: Combining rigorous patient selection (tumor ≤3 cm, no metastases, CA 19-9 ≤100 U/mL), neoadjuvant chemoradiation, and mandatory staging laparoscopy (given 27% incidence of metastases) achieves 5-year post-transplant survival of 65%, comparable to other transplant indications.
- Recent UNOS policy (July 2024) allows MELD exception for solitary iCCA <3 cm in cirrhotic patients with ≥6 months disease stability, based on data showing 73% 5-year survival in highly selected cases.
- The dramatic difference in outcomes between protocol-adherent patients (5-year survival 65%) versus those treated without neoadjuvant therapy (5-year survival 32%) underscores the critical importance of following established guidelines.