Back to Basics: Immunosuppression Fundamentals in Adult Liver Transplantation
Learning objectives:
By the end of this post, the learner will be able to:
- Explain how allorecognition and T-cell activation drive liver allograft rejection.
- Map the major immunosuppressive drug classes to specific steps in the T-cell activation pathway.
Why do we need immunosuppression?
Adult liver transplant recipients almost universally require long-term immunosuppression. Although the liver is relatively ‘tolerogenic’ compared with other solid organs, an unmodified alloimmune response can still produce T-cell mediated hepatocellular and biliary injury, sustained inflammatory damage, progressive fibrosis, and ultimately graft dysfunction and loss.
Immunosuppressive management therefore aims to balance two competing priorities:
1. Prevent rejection by disrupting key steps in the alloimmune cascade, including allorecognition, T-cell activation, and clonal expansion.
2. Minimize over-immunosuppression and drug toxicity.
What are the steps of T-cell activation?
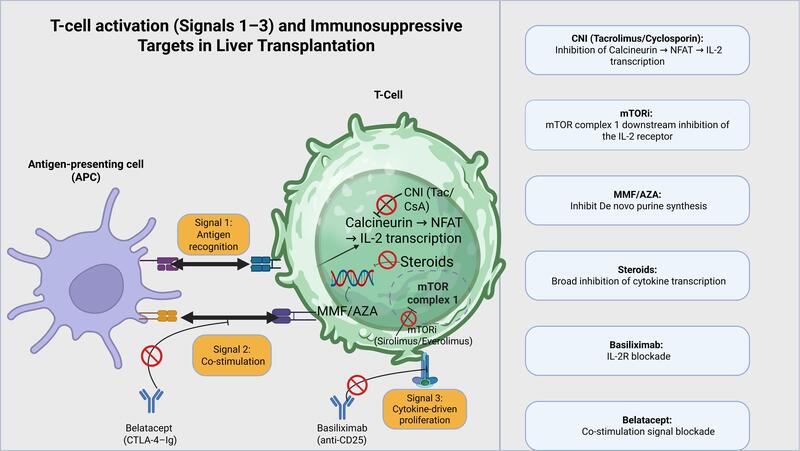
A simple way to organize liver transplant immunosuppression is the three-signal model of T-cell activation, which also serves as a map of where the major drug classes act (Figure 1).
- Signal 1 – Antigen recognition
An antigen-presenting cell (like a dendritic cell or macrophage) presents a processed antigen fragment on its surface via the Major Histocompatibility Complex (MHC) molecule. The T-cell receptor (TCR) on the T-cell recognizes and binds to this specific peptide-MHC complex, delivering the first signal.
- Signal 2 – Co-stimulation
For full activation, the T-cell needs a second signal from co-stimulatory molecules on the APC. CD28 on the T-cell binds B7 proteins (CD80/CD86) on the APC, providing the co-stimulatory signal and amplifying TCR signaling. This increased signaling drives calcium influx and activates the immunophilin–calcineurin pathway: tacrolimus binds FKBP12 and cyclosporine binds cyclophilin, and these drug–immunophilin complexes inhibit calcineurin. Inhibiting calcineurin prevents NFAT activation and reduces IL-2 transcription, which blunts downstream T-cell expansion. Without this second signal, T-cells may become anergic despite antigen recognition.
- Signal 3 – Cytokine-driven proliferation
Successful delivery of Signals 1 and 2 leads to the T-cell producing cytokines, especially Interluekin-2 (IL-2). Intracellular pathways activate calcineurin, which promotes Nuclear Factor of Activated T-cells (NFAT)-mediated transcription of IL-2. IL-2 binds the IL-2 receptor (CD25) and triggers downstream signaling through mTOR and other kinases, leading to clonal expansion and differentiation of effector T-cells that mediate graft injury.
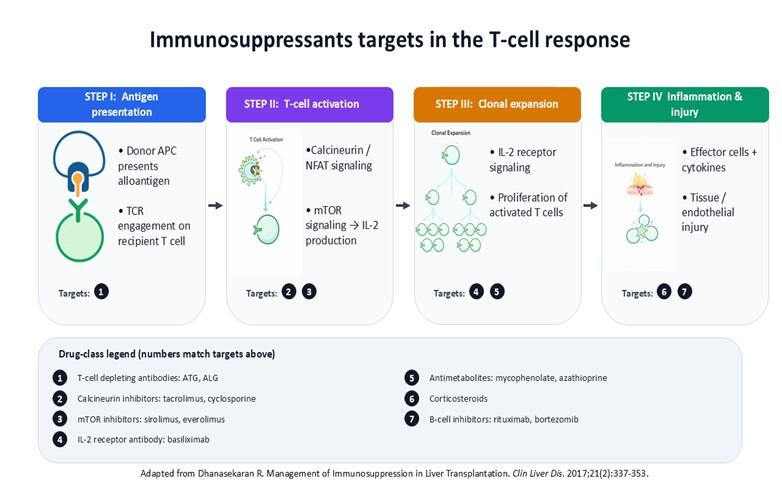
Figure 2: T-cell activation and immunosuppressive targets
Each major immunosuppressive drug class maps onto one or more of these signals:
- Calcineurin inhibitors (tacrolimus, cyclosporine) target signal 1 from the TCR, by inhibiting the phosphatase calcineurin, whose activation promotes nuclear translocation of NFAT and IL-2 transcription (Signal 1, early activation).
- Belatacept, a CTLA-4–Ig fusion protein, blocks CD80/86–CD28 interaction and thereby co-stimulation (Signal 2).
- Basiliximab, a chimeric monoclonal antibody, binds IL-2 receptor alpha chain (CD25), effectively blocking the IL-2R. This prevents IL-2 from attaching to its receptor, and without IL-2 signaling, activated T-cells cannot proliferate (Signal 3).
- The mTOR pathway (mechanistic Target of Rapamycin) is a central cellular signaling network, integrating signals to control cell growth, metabolism, proliferation and protein synthesis. mTOR inhibitors (sirolimus, everolimus) inhibit mTOR complex 1 downstream of the IL-2 receptor, blocking cell-cycle progression (Signal 3).
- Antimetabolites such as mycophenolate and azathioprine impair de novo purine synthesis and limit proliferation of activated lymphocytes.
- Corticosteroids broadly suppress transcription of pro-inflammatory cytokines and promote lymphocyte apoptosis, influencing multiple points in the pathway.
How does pharmacokinetics affect drug behavior?
Calcineurin inhibitors
CNIs have a narrow therapeutic index. They are metabolized largely via CYP3A4 (and CYP3A5) and are substrates for P-glycoprotein (a crucial efflux pump that pumps substances back into the intestinal lumen). Hence bioavailability and clearance are highly sensitive to gastrointestinal factors (food effects, edema, diarrhea), hepatic function, and drug-drug or food-drug interactions.
Antimetabolites
Mycophenolate mofetil and enteric-coated mycophenolate sodium are prodrugs that are hydrolyzed to the active metabolite, mycophenolic acid (MPA). MPA selectively inhibits inosine monophosphate dehydrogenase (IMPDH), thereby limiting de novo guanine nucleotide synthesis.
mTOR inhibitors
Sirolimus and everolimus are mTOR inhibitors that bind the intracellular immunophilin FKBP12, but unlike calcineurin inhibitors act downstream of IL-2 signaling by inhibiting mTOR. In doing so, they blunt IL-2 mediated cell-cycle progression (G1 → S phase), thereby limiting the clonal expansion of activated T lymphocytes.
Both agents are substrates for CYP3A4 and P-glycoprotein, making their exposure susceptible to drug-drug interactions and variability in intestinal/hepatic metabolism.
Corticosteroids
Corticosteroids bind the glucocorticoid receptor and alter gene transcription, decreasing production of pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α while promoting lymphocyte apoptosis.
Co-stimulation blockade
Belatacept is a CTLA-4–Ig fusion protein that binds CD80/86 on antigen-presenting cells, preventing interaction with CD28 on T-cells and thereby blocking co-stimulation (Signal 2).
In liver transplant recipients, it remains reserved for selected scenarios or clinical trials because of concerns about early rejection and post-transplant lymphoproliferative disorder, particularly in EBV-seronegative patients.
For a practical, case-based overview of immunosuppressive regimens and toxicity profiles, see our LFN Clinical Pearls post “A Beginner’s Guide to Liver Transplant Immunosuppression” here.
Table 1. Common immunosuppression agents used in liver transplant recipients
| Class / Drug | Main target / pathway |
| Tacrolimus, cyclosporine | Calcineurin–NFAT; inhibits IL-2 transcription |
| Mycophenolate mofetil, mycophenolate sodium | IMP dehydrogenase; inhibits de novo guanine synthesis |
| Azathioprine | Inhibits de novo purine synthesis → Halts DNA/RNA production |
| Sirolimus, everolimus | mTORC1; inhibits IL-2–driven proliferation |
| Corticosteroids | Glucocorticoid receptor; prevents cytokine gene transcription |
| Belatacept | CD80/86–CD28 co-stimulation |
How should you interpret drug levels across phases of therapy, and what defines a “true trough”?
For most drugs, the time to reach steady state is four to five half-lives if the drug is given at regular intervals. A true trough is ideally drawn before the next dose after steady state is reached. For instance, the half-life of tacrolimus is approximately 12 hours, four to five half-lives correspond to about 2–3 days. A trough drawn the next morning after a major dose change may not reflect the final steady-state level; the most informative “true trough” is the pre-dose level obtained after ~48–72 hours on the new regimen. Troughs approximate overall exposure but are influenced by timing of the blood draw, adherence, food, cholestasis, liver function, and drug-drug interactions through CYP3A and P-glycoprotein.
For a practical, case-based take on CYP3A inhibitors/inducers and tacrolimus level surprises, check out the LFN Hepatology Practice Questions (Edition 1) (drug–drug interaction scenarios)here.
How does immunosuppression affect malignancy risk and immune surveillance?
Global T-cell suppression with calcineurin inhibitors and corticosteroids impairs cytotoxic responses against tumor antigens and facilitates oncogenic viral reactivation. mTOR inhibitors uniquely inhibit tumor cell proliferation and angiogenesis, providing a rationale for their use in patients with high oncologic risk.
Co-stimulation blockade with belatacept interrupts CD28-mediated T-cell activation by preventing the CD80/86-CD28 interaction. By attenuating T-cell activation and downstream T-cell help to B cells, this strategy can alter humoral immune responses. Clinically, belatacept has been associated with an increased risk of post-transplant lymphoproliferative disorder (PTLD), particularly in EBV-seronegative recipients highlighting that targeting different nodes of the T-cell activation pathway can yield distinct downstream effects, including differing oncologic risk profiles.
Check out our LFN Clinical Pearls post “PTLD: A Balancing Act of Immunosuppression” here for a real-world example of how clinicians calibrate immunosuppression when malignancy and infection risk collide.
Take-home points
- Liver transplant immunosuppression is best understood through the three-signal model of T-cell activation, with each major drug class mapping onto at least one signal.
- Calcineurin inhibitors and mTOR inhibitors have a narrow therapeutic window; steady state, true troughs, and CYP3A/P-glycoprotein interactions are central to interpreting drug levels.
- Long-term immunosuppression reshapes infection and malignancy risk, and mTOR inhibitors and co-stimulation blockers have distinct oncologic profiles compared with calcineurin inhibitors and corticosteroids.
Written by: Ahmed El Sabagh, MD, MedStar Georgetown/Washington Hospital Center
Post reviewed by Vinay Jahagirdar (Fellow Lead), Bilal Khalid (Faculty Editor)