Hep, Hep, Hepatitis!
What is HBV reactivation?
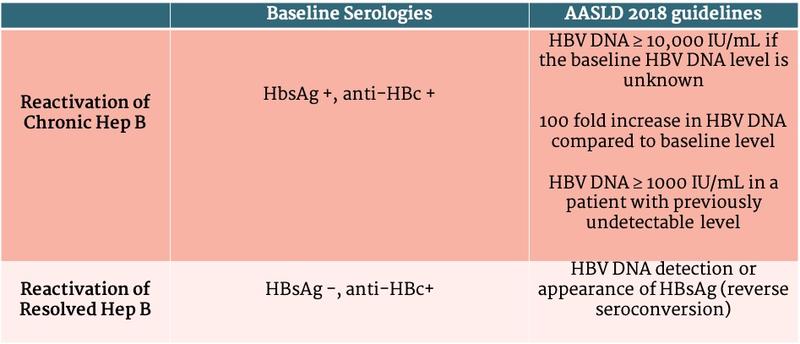
In patients with chronic HBV (defined as HBsAg+ for at least 6 months and measurable HBV DNA in the blood), reactivation is defined by an increase in HBV DNA above baseline whereas in patients with resolved HBV (defined by HBsAg - and anti-HBc+), reactivation is defined by either appearance of HBV DNA in the blood or conversion to HbsAg + state (i.e. reverse seroconversion) (Table 1).
Table 1 - Guideline Specific Definition of HBV Reactivation
Risk factors for reactivation of HBV infection
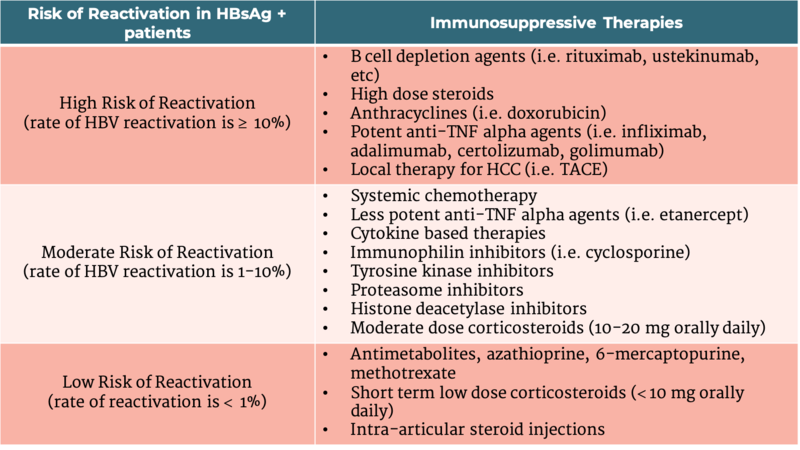
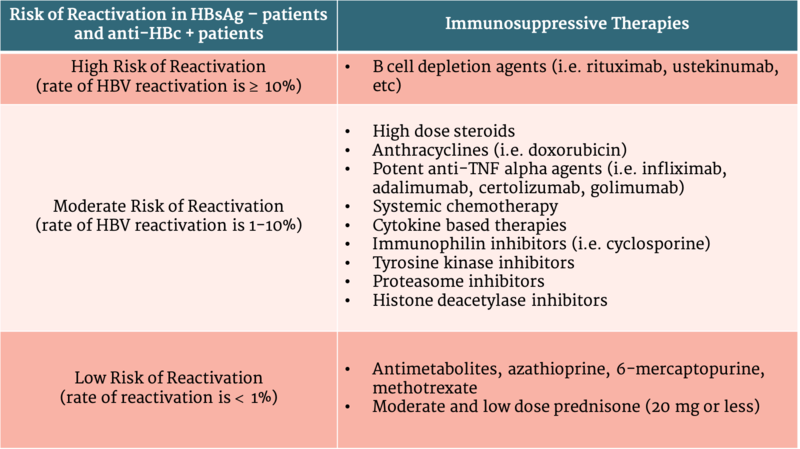
Patients with serologic evidence of HBV infection, manifested by either a HBsAg + or anti-HBc +, are at risk of reactivation, particularly if they receive immunosuppressive therapy. This includes those treated for malignancy or an autoimmune disorder, and those undergoing solid or hematopoietic stem cell transplantation (HSCT). 41%-53% of HBsAg +, anti-HBc + patients compared to 8% -18% of HBsAg -, anti-HBc + patients undergoing anticancer therapies had HBV reactivation. Other studies showed that HBV reactivation from anti-rheumatic therapies was 12.3% in HBsAg + and anti-HBc + patients compared to 1.7% in HBsAg -, and anti-HBc + patients. Therefore, while other risk factors such as male gender and age have been shown to play a role in HBV reactivation, the level of risk is mostly dependent on HBsAg status and the type of immunosuppression used. (Table 2)
Table 2 - HBV reactivation risk stratification for those undergoing immunosuppressive therapy
Approach to screening
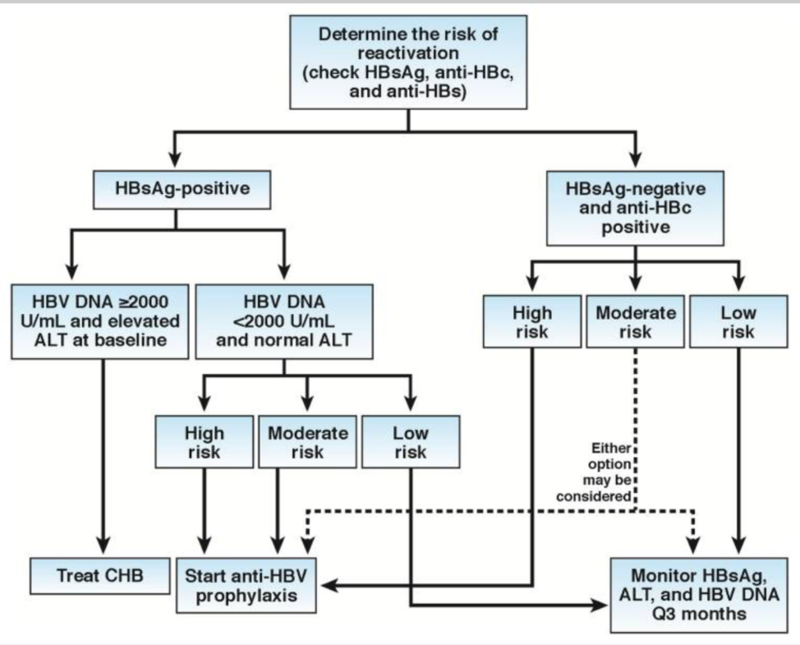
Several guidelines endorse testing for active and chronic HBV in any patient needing immunosuppressive therapy. Risk for HBV reactivation can be readily identified by testing for HBsAg and anti-HBc (Figure 1).
Figure 1 - Approach to HBV testing
Clinical Manifestations of HBV reactivation
Different degrees of HBV reactivation may occur. Silent HBV reactivation is seen with an increase in HBV DNA levels without an increase in aminotransferase levels. Mild reactivation can occur with a rise in serum aminotransferase levels without jaundice or other symptoms, whereas jaundice and/or symptoms of liver injury is seen in moderate HBV activation. Finally, severe HBV reactivation is marked by features of liver failure, which could lead to death.
Preventing HBV reactivation
In general, antiviral therapy initiated prior to immunosuppressive therapy or concurrently can minimize the risk of HBV reactivation. If antiviral therapy is initiated after a flare of hepatitis has already occurred, it may take several months to control the disease and reduce the viral load.
Initially, lamivudine was the first agent to become available for treatment or prevention of HBV reactivation. While safe and generally well tolerated, resistance to viral variants is often seen after prolonged use. As a result, antiviral drugs such as tenofovir and entecavir have now become the first line agents to treat HBV.
The decision to administer prophylactic therapy depends on the level of risk as determined by serologic status and type of immunotherapy administered (Table 2 and Figure 1). Although there is no data to dictate how long therapy is required, most experts recommend continuing antiviral therapy for the duration of chemotherapy, with some recommending continued therapy for 3 to 6 months after the last cycle of chemotherapy while other favor continuing for up to 12 months (especially when anti-CD20 is used).
Various guidelines propose slightly different nuanced approaches (Table 3).
Table 3: Guidelines specific management approaches to HBV Reactivation