The neglected viral hepatitis
What is hepatitis E?
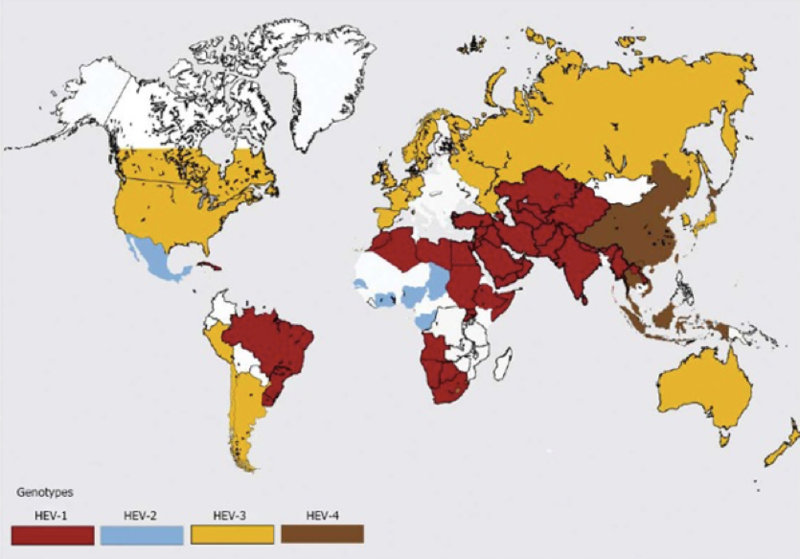
Hepatitis E is a non-enveloped single-stranded RNA virus in the family hepeveridae. It is one of the most common causes of acute viral hepatitis, yet one of the least diagnosed. Hepatitis E is relatively widespread in the developing world, but rare in developed countries. Hepatitis E has four geographically distinct phenotypes. Genotypes 1 and 2 are human viruses that cause epidemic hepatitis, primarily in developing countries, with waterborne and fecal-oral routes of transmission.Genotypes 3 and 4 are swine viruses common in pigs, both in developed and developing countries, with foodborne routes of transmission.
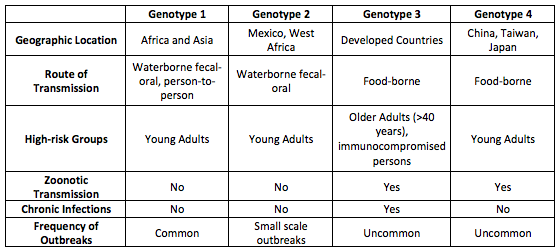
Table adapted from: Centers for Disease Control and Prevention. www.cdc.gov/hepatitis/index.htm
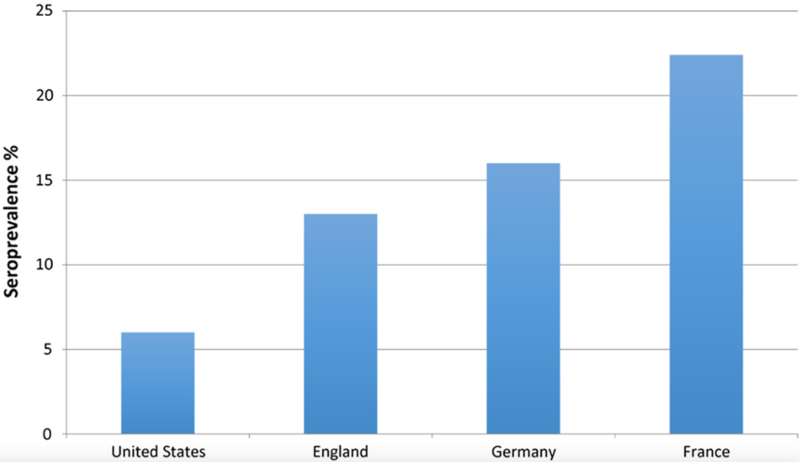
Estimates of the seroprevalence of HEV vary widely, and the distribution is uneven based solely on differences in the source of infection which varies by HEV genotype.
Hepatitis E virus (HEV) most commonly causes a self-limited acute illness. The vast majority of patients (90-95%) with acute HEV are asymptomatic.When symptoms are present, they are typically mild and include malaise, fatigue and anorexia. More severe symptoms include nausea, vomiting, fever, jaundice, abdominal pain and hepatomegaly. Symptoms often coincide with marked elevations in AST and ALT levels to the 1000’s with milder elevations in total bilirubin and alkaline phosphatase.
Most patients (90-99%) who acquire HEV, spontaneously clear the virus and no treatment is required.However, complications such as acute hepatic failure, cholestatic hepatitis and chronic HEV can develop. In regions where HEV is endemic, acute hepatic failure occurs more frequently in pregnant women and is associated with mortality rates of 15-25%. HEV infection can also lead to decompensation in patients with preexisting liver disease and those who are malnourished. Nearly all patients who develop chronic HEV infection are solid organ transplant recipients or are immunosuppressed.
When should chronic hepatitis E be suspected?
Chronic HEV is defined as detectable HEV RNA in serum or stool for longer than 6 months. Chronic HEV infection occurs almost exclusively in immunocompromised patients including patients receiving chemotherapy, organ-transplant recipients and HIV-infected persons. Chronic HEV infection occurs most commonly with genotype 3 infection but cases of chronic HEV have been described with genotype 4, as well.
The source of HEV infection is often unknown but in rare cases, blood transfusions or the transplanted organ itself have appeared to be the source. Europe has seen a 10-fold surge in the number of HEV infections over the past 10 years with the majority secondary to genotype 3, the same genotype isolated in European pigs.The preponderance of cases have occurred in France, Germany and the UK where fresh pork is widely consumed. In contrast, the prevalence of HEV appears to be declining in the United States where processed pork is commonplace.
Symptoms of chronic HEV infection are often minor and non-specific until progression to decompensated cirrhosis occurs. Patients with chronic HEV have minor, persistent elevations in aminotransferase levels with detectable HEV RNA in the serum and stool. Liver biopsy reveals histologic findings compatible with chronic viral hepatitis including focal necrosis, acidophilic degeneration and lobular inflammation composed mostly of lymphocytes.The histologic features overlap with other liver diseases and make the histopathologic diagnosis of hepatitis E challenging.
How do you diagnose hepatitis E?
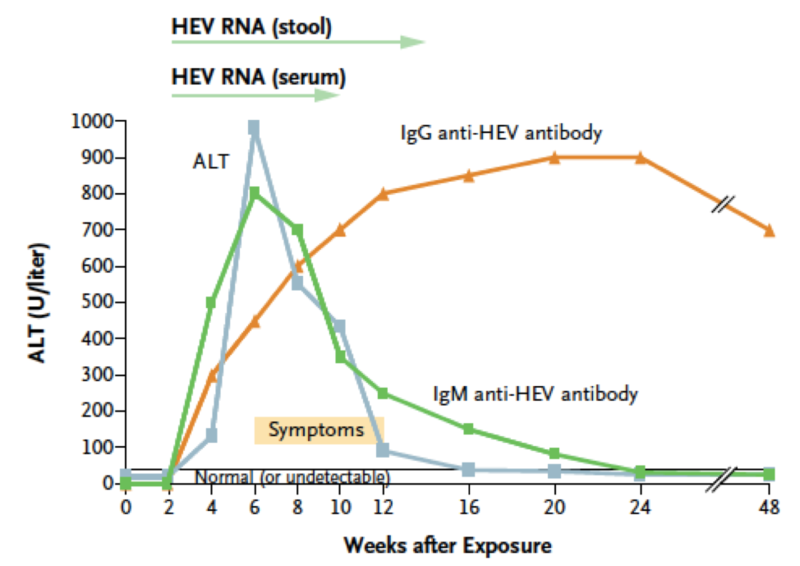
As with other viral hepatitis infections, hepatitis E virus levels rise during the incubation period. Antibodies (both IgG and IgM) appear at the time of clinical onset, just preceding a sharp rise in aminotransferase levels and the development of symptoms. Aminotransferase levels generally return to normal within 6 weeks of the onset of illness, except in cases of chronic hepatitis E.
Image taken from Hoofnagle JH, Nelson KE, Purcell RH. Hepatitis E. NEJM. 2012;367(13):1237-1244.
The diagnosis of HEV should be considered in patients with acute or chronic hepatitis that cannot be explained by other causes. The diagnosis of HEV is complicated by a lack of standardized assays. Tests for anti-HEV antibodies (both IgG and IgM) are commercially available, but the sensitivities and specificities vary widely. In addition, the decline in IgG anti-HEV titers with time may adversely impact sensitivity.In immunocompromised patients, anti-HEV antibodies are often undetectable. In these patients, nucleic acid amplification techniques (NATs) for HEV RNA in the serum or stool may be the only reliable means of diagnosis. In immunocompetent patients, testing for anti-HEV IgM and/or IgG can be considered with HEV RNA testing to confirm the diagnosis. If anti-HEV IgM and IgG testing is negative but clinical suspicion remains high, HEV RNA testing should be considered.
What is the treatment for chronic hepatitis E?
Chronic HEV infection occurs almost exclusively in immunocompromised patients and the mainstay of treatment involves a reduction in immunosuppressive therapy. There is no clear data on how immunosuppressive therapy should be reduced, but for solid organ transplant recipients, a reduction in Tacrolimus is typically recommended first. Clearance of chronic HEV infection has been reported in approximately 30% of patients with a reduction in immunosuppressive therapy alone.
If immunosuppressive therapy cannot be reduced or is unsuccessful in eradicating HEV infection, a 12-week course of ribavirin monotherapy is recommended. A complete blood count should be drawn at weeks 4, 8 and 12 of ribavirin therapy to evaluate for anemia associated with ribavirin. If anemia is to develop, the dose of ribavirin should be adjusted based on severity. Ribavirin is also a potent teratogen and should not be used in pregnant women. For women of childbearing age, pregnancy testing should be performed prior to treatment and 6 months following. Both men and women should be counseled on the use of contraception.
The goal of chronic HEV treatment is to achieve a sustained virologic response (SVR). SVR is defined as undetectable HEV RNA levels 12 weeks after the cessation of treatment. For those receiving ribavirin therapy, both serum and stool HEV RNA is recommended at week 12 of therapy.Ribavirin can be stopped if HEV RNA is negative in both the serum and stool with repeat HEV RNA testing 12 weeks following therapy to determine if virologic response is sustained. There is no viral reservoir for hepatitis E so those patients who achieve an SVR are considered cured, but at risk for reinfection. If HEV RNA testing is positive at week 12, ribavirin therapy should be continued for another 12 weeks. No randomized controlled trials have evaluated the efficacy of ribavirin but case series have demonstrated clearance rates as high as 95%.
Patients who fail to achieve an SVR should continue to be followed for signs of progressive liver disease. There are no established alternative antiviral therapies for treatment failure, but pegylated interferon-alphaand sofosbuvir have been suggested.