Endoscopic Management of Gastric Varices
What Are Gastric Varices?
Gastric varices are vessels in the mucosa and submucosa of the stomach comprising a group of vascular shunts between portal circulation and systemic circulation, typically from prehepatic portal hypertension. They are seen in 17-25% of patients with cirrhosis. While gastric varices bleed less frequently than esophageal varices, gastric variceal bleeding is associated with higher severity, rebleeding risk and mortality. Gastric varices may also have bleeding at lower portal pressures, due to left-sided circulation from a gastrorenal shunt.
Besides portosystemic shunts, gastric varices may also be caused by splenic vein thrombosis, seen in pancreatitis or pancreatic adenocarcinoma, given elevated pressures in the splenic vein. In addition, gastric varices can be due to portal vein thrombosis, even in the absence of cirrhosis.
Classification of Gastric Varices
Gastric varices can be classified based on endoscopy or cross-sectional imaging. Endoscopically, the traditional Sarin classification differentiates between gastroesophageal varices (GOV) and isolated gastric varices (IGV). GOV1 are a continuation of esophageal varices that extend 3-5 cm below the gastroesophageal junction into the lesser curvature of the to stomach. They comprise about 80% of gastric varices.
GOV2 are also a continuation of esophageal varices but extend into the gastric fundus, and IGV1 are gastric varices isolated in the gastric fundus. They are both associated with portal hypertension due to cirrhosis, hepatocellular carcinoma (HCC), or portal vein thrombosis (PVT). However, IGV1 can also be due to splenic vein thrombosis and splenomegaly. GOV2 and IGV1 comprise the true gastric varices known as cardiofundal varices, which make up about 20% of gastric varices. Sarin has identified cardiofundal varices as higher risk for bleeding.
Lastly, IGV2 comprise 1% of all gastric varices. While data is limited, they are considered part of ectopic varices, since they form outside the esophagus and proximal stomach.
The American Gastroenterological Association (AGA) has proposed an alternative nomenclature for gastric varices (Figure 1), based on endoscopic location that also helps correlate with vascular imaging, simplifying gastric varices as lesser curve (GOV1), cardiofundal (GOV2, IGV1) and distal (IGV2).
Endoscopic classification should not be used to guide primary prophylaxis for gastric variceal bleeding. Instead, estimated variceal size and high-risk stigmata, such as platelet plugs, should be documented in addition to the Sarin classification.
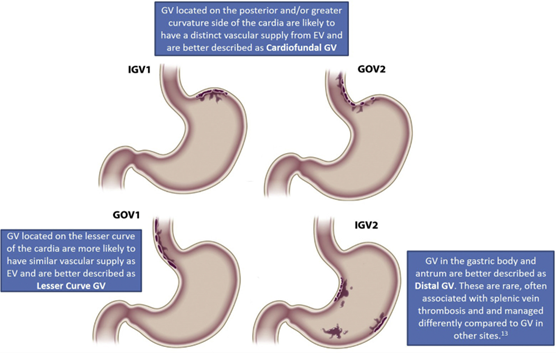
Figure 1: Endoscopic Classification System for Gastric Varices
Since vascular anatomy may differ, cross-sectional imaging with portal venous phase is recommended in the setting of gastric varices. This can characterize the afferent and efferent vascular variations of portosystemic shunts in order to plan more definitive endovascular therapeutics for gastric varices, such as Transjugular Intrahepatic Portosystemic Shunt (TIPS) and Retrograde Transvenous Obliteration (RTO).
Prevention of Initial Gastric Variceal Bleeding
Since gastric varices are characteristic of clinically significant portal hypertension (CSPH), non-selective beta-blockers should be considered as primary prophylaxis in the setting of compensated cirrhosis to prevent decompensation by decreasing cardiac output and causing splanchnic vasoconstriction to decrease portal pressures.
As gastric varices are seen more in prehepatic causes of portal hypertension such as splenic vein thrombosis causing sinistral or left-sided portal hypertension, contrast-enhanced cross-sectional imaging should be obtained to evaluate for thrombosis. For example, patients with noncirrhotic portal hypertension may be better treated with splenectomy if found to have splenic vein thrombosis, which may be overlooked if evaluated only with endoscopy.
If non-selective beta-blockade is contraindicated or poorly tolerated, and if high-risk fundal varices are present, including size greater than 10 mm, red wale sign, Child-Turcotte-Pugh (CTP) class B or C, then Endoscopic Cyanoacrylate Injection (ECI) can be considered for prevention of initial gastric variceal bleeding. Endoscopic Ultrasound (EUS)-guided techniques have also be utilized for primary prophylaxis with small rebleeding rates. Of note, neither TIPS or RTO are recommended for prevention of initial bleeding for gastric fundal varices given lack of data.
Initial Management of Acute Gastric Variceal Bleed
Patients who present with acute bleeding from gastric varices can be initially medically managed similar to an esophageal variceal bleed. This includes resuscitation with a restrictive hemoglobin goal of 7-9 g/dL and antibiotic therapy such as ceftriaxone 1 g daily for maximum 7 days. While there is limited data, vasoactive medications such as octreotide and terlipressin have been used as well.
Endoscopy should be pursued within 12 hours of presentation with the goal of identifying the source of bleeding, classifying the site, and achieving acute hemostasis for hemodynamic stabilization in order to plan on further diagnostic evaluation, Patients should be transferred to a specialized center if needed.
Hemostatic therapies should be utilized based on available resources and local expertise. Endoscopic sclerotherapy with alcohol-based agents such as ethanolamine are not recommended unless there are no other options, given high incidence of rebleeding and risk of developing deep ulcerations. Band ligation can be a reasonable temporizing therapy for cardiofundal varices, only if followed by more definitive management. Endoscopic cyanoacrylate injection (ECI) is another option for initial hemostasis; however, its logistics may restrict its usage during the initial diagnostic endoscopy.
If unable to achieve hemostasis endoscopically, balloon tamponade with inflation of only the gastric balloon can be used, such as Sengstaken-Blakemore or Linton-Nachlas tube. Since the balloon tension may cause mucosal breakdown, it should only be used to swiftly bridge to other definitive therapies.
Overview of Definitive Management for Prevention of Rebleeding Gastric Varices
A multidisciplinary discussion should take place between hepatology, interventional radiology and advanced endoscopy in order to best define definitive management of gastric varices based on endoscopic appearance, vascular anatomy as well as other complications of portal hypertension.
Given similarities, GOV1 should be managed similarly to esophageal varices. Bleeding from fundal varices or distal gastric varices can be managed with either ECI, TIPS or RTO if TIPS is contraindicated. Pending local expertise, and if hemodynamically stable, ECI or EUS-guided coil injection can be utilized. If unavailable or if hemodynamically unstable without otherwise contraindications, TIPS or RTO can be alternatively pursued.
Lastly, if bleeding is due to isolated splenic vein thrombosis, patients should be evaluated for splenectomy, splenic vein stenting or splenic artery embolization.
Endovascular Management of Gastric Varices
TIPS is an effective method to reduce portal pressure and favored in patients with other complications of portal hypertension, such as ascites and large esophageal varices. It can be utilized when lesser curvature gastric varices are refractory to band ligation therapy. When able, TIPS should be combined with RTO or direct endoscopic embolization for gastric varices for better control of bleeding, especially if still present on follow-up EGD. Risks of TIPS include hepatic encephalopathy and hepatic ischemia, which may be greater in the presence of a gastrorenal shunt often associated with cardiofundal varices.
RTO, such as balloon occlusion (BRTO), coil assisted (CARTO) or vascular plug assisted (PARTO), can be a good option if TIPS is contraindicated and preferred in patients with overt hepatic encephalopathy or poor liver function. It requires the presence of a gastrorenal shunt. Risks of RTO include complications of portal hypertension, which may worsen given the subsequent increase in portal pressures. Refer to the LFN article on RTO for a more detailed discussion on these techniques. Follow-up EGD and cross-sectional imaging should be obtained to confirm eradication of gastric varices and evaluate for vascular complications.
Endoscopic Cyanoacrylate Injection
While the large formation of cardiofundal varices makes band ligation challenging, Endoscopic Cyanoacrylate Injection (ECI) is an effective method to prevent rebleeding of gastric varices, especially if there is no gastrorenal shunt present. This involves directly visualizing and injecting acrylic resin monomers that undergo rapid chain polymerization when in contact with hydroxide ions, forming strong solid chains that cause thrombosis. ECI should be performed with interventional radiology available in case of complications.
The most commonly used material is cyanoacrylate (CA), where 4 carbon (butyl) such as Histoacryl can polymerize faster than 8 carbon (octyl) such as Dermabond, which is more readily available. Faster polymerization can reduce the risk of embolization and bleeding. Plant-based oils like lipiodol are radiopaque and had previously been used for radiographic confirmation; however, it may delay polymerization and increase embolization risk and therefore no longer recommended.
Set up includes preparing 2 mL syringes with 1 mL glue and 1 mL distilled water, and normal saline should be avoided since this delays polymerization. In addition, a 23G or 21G needle is used for the injection and acetone is set aside in case of glue spillage from the endoscope channel.
During an acute gastric variceal bleed, the needle is placed into the varix adjacent to the actual bleeding site. Distilled water is flushed first, then 1 mL of glue is injected followed by 1 mL distilled water, which prevents needle blockage. If bleeding persists or the varix is soft when palpated, further injections can be given until the varix solidifies (Figure 2). In addition, any adjacent varices should be injected until obliteration. For gastric varices not actively bleeding, injections should start peripherally until the visible varix and its tributaries are solidified. At least 10 seconds should pass until withdrawing the needle in order to prevent endoscope channel blockage.
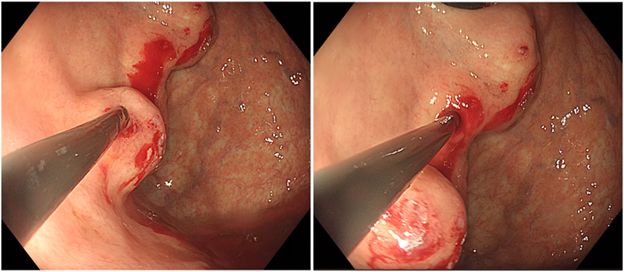
Figure 2: Endoscopic Cyanoacrylate Injection
The most reported complication is embolization of glue causing pulmonary embolus or stroke, seen incidentally with lipiodol uptake in the lungs, which can occur at a rate of 0.7%. Other complications include needle impaction in the gastric varix and inducing gastric variceal bleeding, which can be avoided with good technique. Portal and splenic vein thrombosis are really rare, and infections in active bleeding should be prevented with empiric antibiotics in the setting of variceal bleeding.
If ECI is utilized as main definitive therapy, non-selective beta-blockers should be added if tolerated. Treatment should be repeated every 2-4 weeks until obliteration, then within 3-6 months and subsequently annually for surveillance.
EUS-Guided Coil Injection Therapy
Taking into consideration the multiple risks of ECI, endoscopic ultrasound (EUS) provides new ways that mitigate those risks by using a fine needle aspiration (FNA) needle to place hemostatic coils with an adjunct into gastric varices. Several studies have shown the benefits of EUS-guided coil therapy, including its ability to utilize fewer procedures and less rebleeding episodes compared to direct ECI alone.
Endosonography allows for specific intravariceal targeting with real-time feedback of hemostasis as well as better characterization of variceal size, adjacent feeder vessels and flow via doppler. Given ECI would require a clear direct view to inject fundal varices in a retroflexion position, EUS can target those varices in a straight approach through a transesophageal or transgastric view. The hemostatic coils that promote thrombosis have a soft platinum wire core with spaced synthetic fibers (Figure 3), which act as a scaffold for any subsequent adjunct injection to reinforce and reduce risk of postprocedural systemic embolization. The adjuncts help reinforce the hemostasis of the coils, which include glue or gelfoam.

Figure 3: Hemostatic Coils
After administration of prophylactic antibiotics, about 200 cc of water is placed into the gastric fundus to improve visualization with EUS doppler, which can differentiate between intramural vessels of gastric varices from extramural perforator veins.
Coils are then chosen with diameter based on FNA needle (0.018” coils with 22G needle and 0.025” coils with 19G needle), and length about 30-50% diameter of the varix. Upon needle insertion into the varix, the stylet is advanced to deploy the coils, which is repeated until significant loss of doppler flow or inability to add more coils (Figure 4). Contrast can be used to confirm a low embolization risk. After noted decrease in doppler flow, an adjunctive agent then can be placed, which includes cyanoacrylate, thrombin, absorbable gelatin sponge (Surgiflo, Gelform), RAD16 self-assembling peptide (Purastat). Lastly, fluoroscopy can also confirm coil placement but is not required.
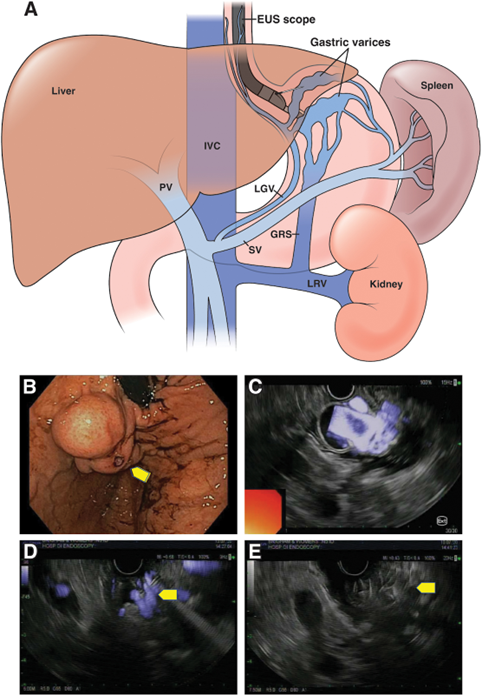
Figure 4: EUS-Guided Coil Therapy with Subsequent Decrease in Doppler Flow
This procedure should be repeated every 2-4 weeks until obliteration with subsequent surveillance similar to band ligation for esophageal varices. For persistent gastric varices, repeat cross-sectional imaging should be obtained to guide other therapies.
Back to the case
Multidisciplinary discussion between hepatology, interventional radiology and advanced endoscopy decided to proceed with EUS-guided coil therapy as secondary prophylaxis for gastric variceal bleed.
Endosonography showed tubal, anechoic, doppler-positive structures in the gastric fundal wall, consistent with known varix, measuring at least 10 mm in cross-sectional diameter. The varix was punctured with a 19G FNA needle under endosonography, then two embolization coils were deployed under endosonographic and fluoroscopic guidance starting distally then working proximally with significant decrease in doppler flow. Then 2-3 cc of diluted absorbable hemostatic gelatin sponge was deployed into the varix and around the coils with complete cessation of doppler flow.
After four weeks, EUS showed smaller but still apparent GOV2, which were again treated with two additional coils and hemostatic gelatin sponge. Subsequent repeat surveillance EUS showed no overt persistent gastric varices.
Conclusions
In summary, bleeding gastric varices are associated with significant risks and mortality. The American Association for the Study of Liver Diseases (AASLD) provides a sample algorithm to the management of gastric varices (Figure 5), which includes resuscitation, stabilization as well as cross-sectional imaging and multidisciplinary discussions on definitive endovascular or endoscopic therapies. EUS-guided coil therapy has been shown to be an effective method of definitive endoscopic management for gastric varices, especially when other initial therapies or endovascular therapies are limited or contraindicated.
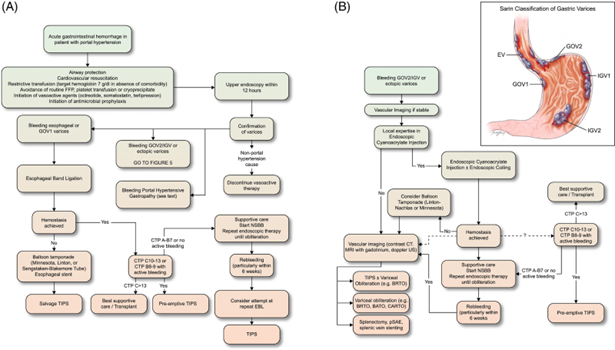
Figure 5: Algorithm for the Management of Acute Variceal Bleeding
Take Home Points:
- Gastric varices should be documented with an estimated variceal size and presence of high-risk stigmata, as well as its endoscopic classification, which includes lesser curve (GOV1), cardiofundal (GOV2, IGV1) and distal gastric varices (IGV2).
- Contrast-enhanced cross-sectional imaging with portal venous phase should be obtained in the setting of gastric varices, to guide definitive management by defining vascular anatomy and evaluate for thrombosis, such as portal vein thrombosis and splenic vein thrombosis.
- Patients who present with acute bleeding from gastric varices should undergo initial medical management and stabilization, followed by endoscopy to identify and stop the source of bleeding.
- After initial hemostasis, a multidisciplinary discussion should take place between hepatology, interventional radiology and advanced endoscopy in order to best define definitive management of gastric varices based on endoscopic appearance, vascular anatomy as well as other complications of portal hypertension.
- EUS-guided coil injection therapy utilizes doppler for real-time assessment and injection of hemostatic coils that act as a scaffold for an adjunctive to mitigate embolization, and can be a good option for definitive management of fundal gastric varices, particularly when not amenable to TIPS or RTO.
